Article by Gina Oller, embryologist and psychologist specializing in fertility and assisted reproduction, in collaboration with Teletest.
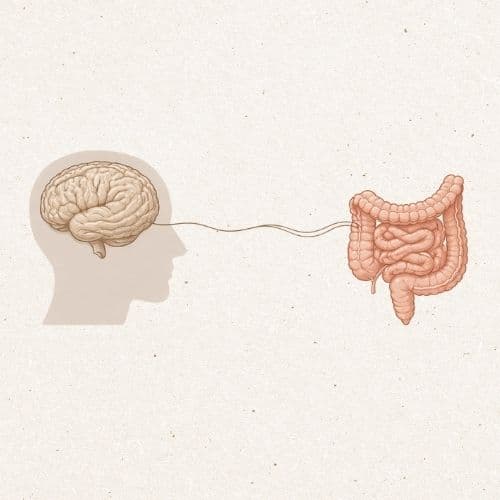
When a couple consulted about fertility, the focus was almost entirely on hormones, ovarian reserve, embryo quality, or seminogram. However, in recent years, another concept has gained weight and is being talked about more and more: low-grade chronic inflammation. This is not an acute inflammation, with pain or clear symptoms. It is a more subtle state, maintained over time, which may be related to the immune system, metabolism, hormonal balance… and also to the gut microbiota.
Although it does not always give obvious signs, this type of inflammation can interfere with sensitive reproductive processes such as ovulation, embryo implantation, or seminal quality. In this article, we explain what the scientific evidence suggests about this relationship and why gut microbiota may be another piece to take into account when seeking pregnancy.
The connection between the gut and fertility
The intestine does not function as an isolated organ. Through the gut microbiota, it is closely related to the immune system, inflammation, and hormonal regulation. When the microbiota loses its balance—known as intestinal dysbiosis—mechanisms associated with a state of persistent, albeit low-intensity, inflammation can be activated. In some cases, this imbalance has been linked to increased intestinal permeability, which allows certain bacterial components to pass into the blood and promotes chronic inflammation.
This type of inflammation does not always produce clear digestive symptoms, but it has been described associated with various health problems, including some contexts of infertility with no apparent cause.
How can inflammation influence female fertility?
In women, low-grade chronic inflammation is described in situations such as polycystic ovary syndrome, endometriosis, or reproductive aging (Orisaka et al., 2023). When this inflammatory state is maintained over time, it can alter the ovarian environment and affect delicate processes such as oocyte maturation, hormonal function, or endometrial receptivity, even when basic hormone tests appear normal. This approach helps to understand why, in some cases, reproductive difficulties may exist without a clear cause in conventional studies.
What about male fertility?
Male fertility does not depend solely on the seminogram. A review published in ‘Frontiers in Microbiology’ describes a possible relationship between intestinal dysbiosis, low-grade systemic inflammation, and male reproductive function (Lv et al., 2024). The authors explain that, in contexts of alteration of the intestinal barrier, the activation of the immune system could indirectly influence the functioning of the testicle, with possible effects on seminal quality and hormone production. Again, this is not a direct cause, but one more factor within a complex context (Lv et al., 2024, Wei et al., 2024).
What are the implications of all this?
With the available evidence, the most prudent thing is to understand gut microbiota and low-grade chronic inflammation as part of a broader context, not as a single cause of infertility (Moustakli et al., 2025; Ameho et al., 2025; Lv et al., 2024, Orisaka et al., 2023, Wei et al., 2024).
Many factors influence fertility: diet, lifestyle, stress, medication, hormonal and metabolic status. The interest in microbiota lies in identifying possible elements that are not always assessed in routine studies and that could contribute to a persistent inflammatory environment.
What can I do?
In practice, exploring this aspect may make more sense when:
- There are persistent digestive symptoms
- There are signs of sustained inflammation
- It is a case of infertility with no apparent cause
Always as part of a global and personalized approach. Current science cannot affirm that correcting intestinal dysbiosis will solve a fertility problem, but it does suggest that knowing the state of the intestine can provide useful information in some specific cases.
How Teletest can help you
One of the tools available to assess this aspect is the Intestinal Dysbiosis Test, which analyzes the composition of the gut microbiota and some markers related to inflammation or alteration of the intestinal barrier.
This test does not diagnose infertility or replace routine gynecological or andrological tests, but it can provide complementary information to better understand the general context of intestinal health. Its results should always be interpreted by a healthcare professional, along with the clinical history and other studies.
In certain cases, having this information can help make more informed decisions within a comprehensive approach to fertility.
REFERENCES
- Moustakli E, et al. Gut Microbiome Dysbiosis and Its Impact on Reproductive Health: Mechanisms and Clinical Applications. Metabolites (MDPI). 2025.
- Ameho S, et al. The effect of chronic inflammation on female fertility. Reproduction. 2025 (PubMed).
- Patel N, et al. Distinct gut and vaginal microbiota profile in women with recurrent implantation failure and unexplained infertility. BMC Women’s Health. 2022.
- Lv S, et al. Gut microbiota is involved in male reproductive function: a review. Frontiers in Microbiology. 2024.
- Orisaka, M., Mizutani, T., Miyazaki, Y., Shirafuji, A., Tamamura, C., Fujita, M., … & Yoshida, Y. (2023). Chronic low-grade inflammation and ovarian dysfunction in women with polycystic ovarian syndrome, endometriosis, and aging. Frontiers in endocrinology, 14, 1324429.
- Wei, M., Liu, H., Wang, Y., Sun, M., & Shang, P. (2024). Mechanisms of male reproductive sterility triggered by Dysbiosis of intestinal microorganisms. Life, 14(6), 694.


 BUDGETS
BUDGETS ACCESS TO YOUR TESTS
ACCESS TO YOUR TESTS MY RESULTS
MY RESULTS PROFESSIONAL ACCESS
PROFESSIONAL ACCESS